Urinary tract infections (UTIs) are common in small animal practice; it has been reported that up to 27% of dogs will develop infection at some time in their lives.1
Most UTIs are successfully treated with commonly used drugs, dosages, and administration intervals. However, infections can be challenging to effectively treat when they involve the kidneys (pyelonephritis) and prostate (prostatitis). In addition, it can be difficult to create an appropriate antibiotic prescription in patients with kidney disease due to reduced drug clearance.
Understanding drug pharmacokinetics (PK) and pharmacodynamics (PD) is essential when determining the most effective antibiotic therapy. In addition, successful antimicrobial therapy requires appropriate choice of antibiotic, including dose, frequency, and duration (Figure 1).
Pathophysiology of UTI
Nearly all infections are caused by pathogenic bacteria, although fungal or viral UTIs may be rarely encountered. Most bacterial lower UTIs result from bacteria ascending the external genitalia and urethra. Less commonly, bacteria travel hematogenously and colonize the urinary tract.
Numerous innate defense mechanisms help prevent a UTI. Complete and regular voiding, along with intrinsic properties of urine (high osmolality, antimicrobial solutes), helps create a hostile environment for microbes within the urinary tract. Anatomic barriers and mucosal defenses further prevent adherence of virulent bacteria to the urothelium.
Pathogenic bacteria increase the permeability of the urothelium, allowing passage of inflammatory solutes into the subepithelium as well as inflammatory cytokine secretion.2 The result is inflammation and pain, which manifest as dysuria, pollakiuria, stranguria, and/or hematuria. Eradication of the virulent organism can allow the normal permeability and integrity of the urothelium to be restored.
Classification of UTI
There are several classifications of UTI:
Uncomplicated UTI is a sporadic bacterial cystitis found in a healthy patient with normal urinary tract anatomy and function.3
Complicated UTI occurs in a patient with functional or anatomic abnormalities of the urinary tract, or in patients with risk factors for persistent or recurrent infection as well as treatment failure. Such conditions include immunosuppression (due to natural disease or prescribed therapy), diabetes mellitus, hyperadrenocorticism, kidney disease, prostatitis, pregnancy, urinary incontinence, and altered neurogenic function of the bladder.4
Recurrent UTI requires investigation to determine whether it is reinfection, relapsing, or refractory.
Reinfection is the return of a UTI—caused by a different organism—within 6 months of discontinuation of antibiotic therapy.
Relapsing UTI occurs when the same organism is cultured again within 6 months of discontinuation of antibiotic therapy. This suggests that the patient has a condition that allows recolonization or one that prevents total eradication of infection; additional diagnostics are warranted in these patients (Table 1).
Refractory UTI occurs when a positive urine culture is obtained during appropriate antibiotic therapy (based on in vitro susceptibility testing). There are several possible causes for refractory UTI, including:
- Decreased renal drug elimination (results in lower than expected urine drug concentration)
- Inappropriate drug dose or administration schedule
- Low drug bioavailability (eg, due to drug compounding or gastrointestinal disease)
- Poor drug compliance
Additionally, some drugs may show efficacy in vitro, but, for unknown reasons, the same effect is not present in vivo.
Diagnostics and Data for Antibiotic Selection
Pharmacokinetics and Pharmacodynamics
PK is the movement of a drug throughout the body and includes absorption, distribution, metabolism, and excretion. PD is the effect of the drug on the body; in the case of antibiotics, this also includes the effect on the microorganism. These relationships, often referred to as PK/PD, help predict the outcome of any drug prescription.
Alterations in PK may happen with abnormal absorption (eg, severe gastrointestinal disease), drug metabolism (eg, synthetic liver dysfunction), altered protein binding (eg, uremia, hypoproteinemia), and diminished drug excretion (hepatic or kidney failure). An antibiotic’s PD is assessed clinically through in vitro culture and susceptibility testing.
Culture and Sensitivity
Ideally, all patients with a suspected UTI should have a urine sample collected via cystocentesis and evaluated by aerobic culture and antibiotic susceptibility testing. Urine culture is considered the gold standard in the diagnosis of UTI.
The 2 techniques for determining antibiotic susceptibility are the disk diffusion and serial dilution methods. Disk diffusion testing is considered less reliable and does not provide the minimum inhibitory concentration (MIC) of the antibiotic, whereas antimicrobial dilution does provide the MIC and is the preferred method of antibiotic susceptibility testing.
With antimicrobial dilution, a concentration of an antibiotic is added to a liquid medium inoculated with the bacterial isolate. The concentration of antibiotic is doubled in each subsequent well. The MIC is the concentration of antibiotic found in the first tube that exhibits no detectable growth,5 and this concentration is used to categorize the isolate as susceptible, intermediate, or resistant (see CLSI Classification).
Rarely, false-negative findings may result from inappropriate urine storage or slow organism growth (as seen with Corynebacterium species). Urine processing for quantitative culture should be performed immediately after urine is obtained because bacterial colony count dramatically drops after 24 hours of refrigerated storage when urine is stored in a silicone clot tube.6 If urine cannot be processed for quantitative culture immediately, use of a urine transport tube is recommended to help prevent false-negative or underestimated colony count results.
Urine Drug Concentration
Many antibiotics are excreted primarily in urine and achieve concentrations substantially higher than those in plasma. The urine drug concentration should be evaluated with respect to the isolate’s MIC to determine the likelihood of eliminating the organism. Table 2 lists observed urine concentration of antibiotics at the specified dosages in healthy animals.
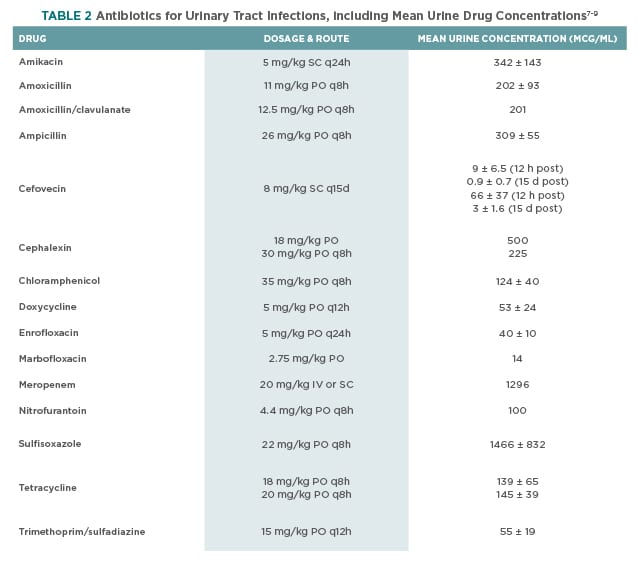
Urine antibiotic concentrations in veterinary patients with kidney disease have yet to be investigated. Reduced glomerular filtration rate (GFR) may decrease the excretion of the drug into urine, resulting in decreased urine concentrations. Additionally, polyuric patients may experience further reduction of urine drug concentration because of increased daily urine volume and subsequent dilution of filtered antibiotic.
Decreased urine excretion caused by diminished GFR can result in plasma drug concentrations exceeding those normally observed and may cause adverse effects. This is most true for drugs with significant kidney elimination. Drugs with mostly hepatic elimination may have minimal alteration in drug excretion in patients with decreased GFR. However, the accumulated uremic toxins and hypoproteinemia present in many patients with kidney disease can lead to altered drug protein binding and abnormal drug PK/PD.
Antibiotic Selection by UTI Classification
Empiric Antibiotic Selection
The emergence of antimicrobial resistance and multidrug resistance has increased, making empiric antibiotic selection very difficult, particularly when previous antibiotic therapy has been administered to the patient.4
Uncomplicated UTI
Recommended drugs for uncomplicated UTI include amoxicillin, cephalosporins, and trimethoprim-sulfonamide.3,6 Although patients with an uncomplicated UTI are often successfully treated empirically, repeated treatment without culture and susceptibility results may lead to incorrect choice of antimicrobial, unnecessary adverse effects, and potential selection of resistant bacteria.4
Complicated & Recurrent UTI
Antibiotics should never be selected empirically for complicated UTI without culture susceptibility results (see Culture & Sensitivity). Management of pyelonephritis, prostatitis, and relapsing or recurrent UTI is often unsuccessful without therapy guided by culture and susceptibility results. However, therapy should be instituted while culture and susceptibility results are being awaited. Rational initial drug choices for complicated UTI include amoxicillin, fluoroquinolones, or trimethoprim-sulfonamide.3
Antibiotic Selection by Urine Drug Concentration
To best use antibiotic urine data, an important consideration is whether a drug is time- or concentration-dependent.
Time-Dependent Drugs
Time-dependent drugs include beta-lactams, cephalosporins, sulfa drugs, tetracyclines, and chloramphenicol. These drug classes are most effective when the tissue concentration exceeds the isolate’s MIC for 50% to 75% of the dosing interval.10 However, few clinical trials have evaluated this suggestion.11
Product inserts and pharmacology texts often contain the drug elimination curves, which are helpful in choosing the dosage and frequency that ensure these criteria are met. The plasma drug elimination curve and renal drug elimination rate can be used as surrogates to predict the urine drug concentration curve.12
Less is known about urine drug concentration and clinical efficacy, but several authors have stated that urine, not plasma, drug concentration is important in ensuring successful eradication of bacteria (see Urine Drug Concentration & Clinical Efficacy).7,9,12
Concentration-Dependent Antibiotics
The efficacy of concentration-dependent antibiotics is best predicted by the Cmax and the isolate’s MIC. Such drugs as fluoroquinolones and aminoglycosides are most effective when the Cmax is at least 8- to 10-fold higher than the MIC.13 These drugs are typically administered every 24 hours.
Another method to evaluate the activity of these antibiotics is comparing the drug concentration area under the curve (AUC) to the MIC. This AUC/MIC ratio has been investigated for some antibiotics, and although some authors generally recommend an AUC/MIC greater than 125 to 250, studies have shown some drugs to be effective with an AUC/MIC of 40.7,14,15
The dosage of these antibiotics is typically chosen to create a high peak urine concentration, well above the isolate’s MIC. Once-daily administration is acceptable for most concentration-dependent drugs, and this frequency may help increase owner compliance in administering medications. However, it may not be consistent with antibiotic stewardship to prescribe a fluoroquinolone antibiotic for an uncomplicated lower UTI when drugs belonging to the penicillin or cephalosporin class would also be effective.
Duration of Therapy
The ideal duration of antibiotic therapy for uncomplicated and complicated UTI is unknown. Many textbooks recommend 10 to 14 days for uncomplicated UTI and 4 to 8 weeks for complicated UTI; however, these guidelines are not evidence-based, and much shorter durations are the standard of care in human medicine.
In 2011, the International Society for Companion Animal Infectious Diseases published recommendations regarding antimicrobial therapy in UTI.3 The recommendations mostly reflect expert consensus because well-designed clinical trials to determine optimal antibiotic duration are lacking in veterinary medicine.
- For uncomplicated UTI, this group recommended 7 or fewer days of antibiotic therapy; humans are typically treated for 3 to 7 days.
- For complicated UTI, the group recommended antibiotic therapy for up to 4 weeks; humans are typically treated for 1 to 2 weeks, although 3 weeks may be indicated in some instances.
Recently, 2 studies evaluated short duration versus long duration of antibiotics for uncomplicated UTI in dogs (3 days of trimethoprim-sulfamethoxazole versus 10 days of cephalexin and 3 days of enrofloxacin versus 14 days of amoxicillin-clavulanic acid).17,18 Both studies demonstrated that the short duration of antibiotic administration was noninferior to the longer duration in bacterial cure rates. However, because both studies compared short duration of one drug with long duration of another, their design precludes determination of optimal treatment time for the drugs investigated.19
A systematic literature review conducted in 2015 to determine the optimal therapy for UTI in veterinary medicine found insufficient evidence available for analysis.19 Currently, evidence-based guidelines for the duration of UTI in small animals do not exist, and further studies evaluating a single drug in both short and long durations of therapy are needed.
Monitoring Response to Therapy
Patients with a simple, uncomplicated UTI may not require rigorous monitoring. However, patients with complicated, relapsing, or recurrent infections should be monitored very closely. The following protocol is recommended to monitor response to therapy in patients with relapsing, recurrent, or refractory UTI.3
- Recheck urine culture 5 to 7 days into antibiotic therapy. This confirms that the prescribed dose and frequency of the drug were successful in treating the organism isolated. This culture also may reveal an additional isolate that could not be identified in the initial culture. Any bacterial growth observed at this time suggests treatment failure. Reconsider the choice of antibiotic, dose, and administration frequency.
- Recheck urine culture 3 days before discontinuing antibiotic therapy. This is an optional step, but it confirms that, when therapy was discontinued, the patient still had a negative culture. Positive bacterial growth at this stage suggests a refractory infection or newly inoculated organism. Investigate patients for any nidus of infection (eg, urolithiasis, anatomic abnormality, local neoplasia). Alter treatment and institute new therapy for the same duration as previously intended.
- Recheck urine culture 7 days after discontinuing antibiotic therapy. Positive growth should prompt investigation for causes of relapse or reinfection.
Complicated, relapsing, recurrent, and refractory UTI may be challenging to cure. However, understanding drug PK/PD and potential alterations in the animal’s metabolism/excretion of the drug can help increase the likelihood of successful treatment.
Summary
Guidelines for appropriate antibiotic dosing for animals with kidney disease have not been established; therefore, a working knowledge of pharmacology and the prescribed drug’s PK/PD profile is needed to help create a successful antibiotic prescription with the smallest risk for adverse effects.
When possible, in patients with kidney disease, avoid drugs that have a narrow margin of safety and undergo significant renal elimination (eg, fluoroquinolones in cats, aminoglycosides) and then choose alternative drugs (based on susceptibility results) that undergo hepatic elimination or those with a wide margin of safety.
Author’s Note